Effectiveness of the Pfizer-BioNTech and Oxford-AstraZeneca vaccines on covid-19 related symptoms, hospital admissions, and mortality in older adults in England: test negative case-control study | The BMJ

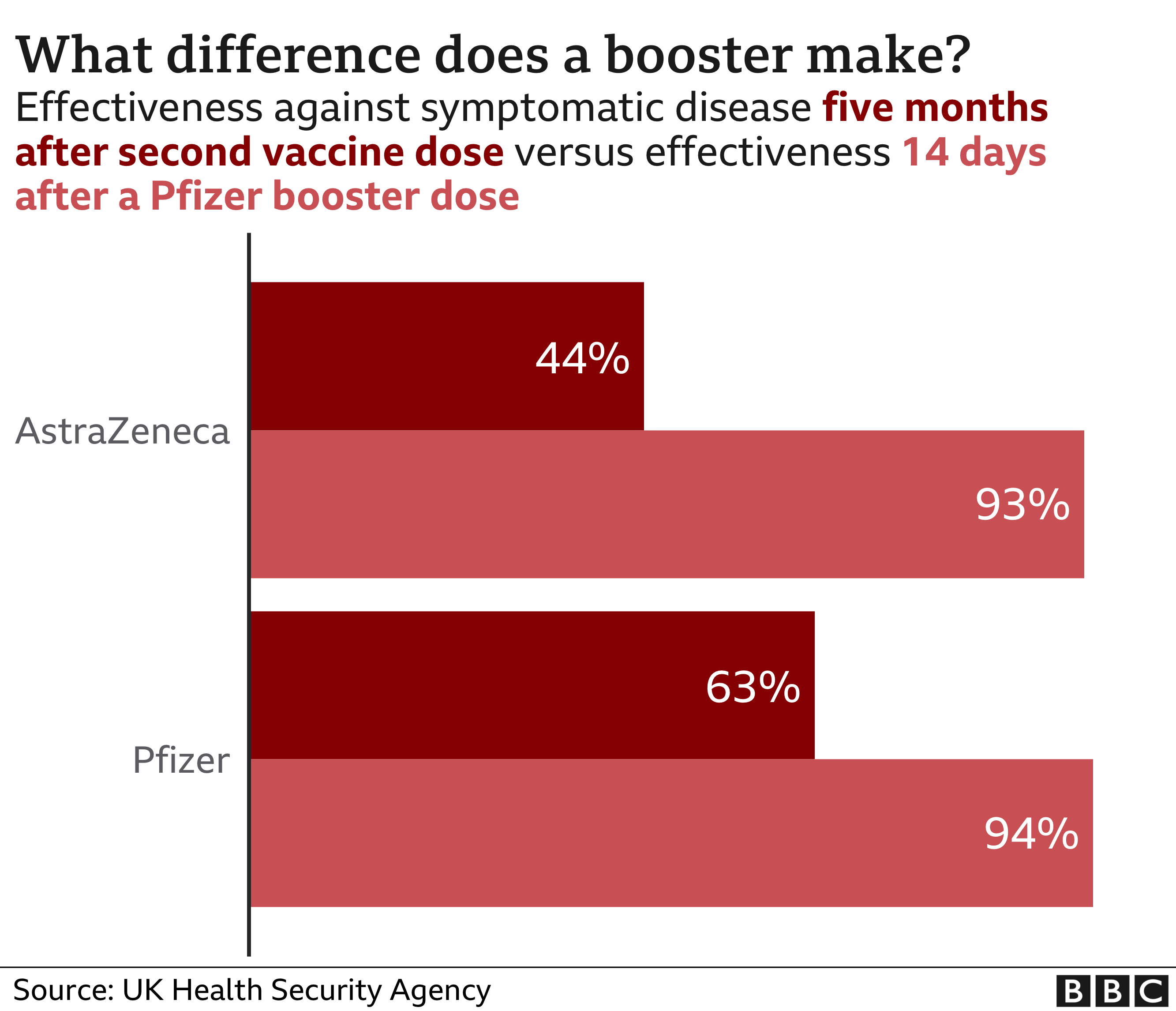
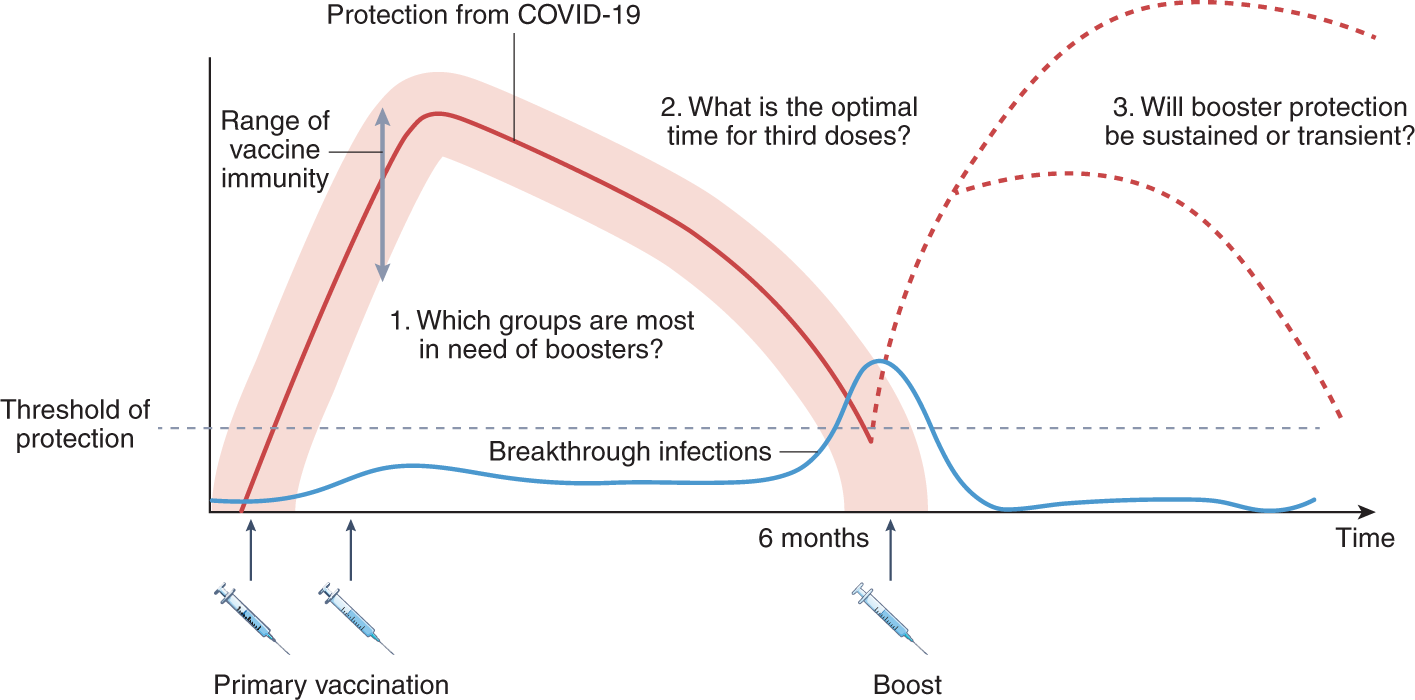
COVID-19 vaccine waning and effectiveness and side-effects of boosters: a prospective community study from the ZOE COVID Study - The Lancet Infectious Diseases

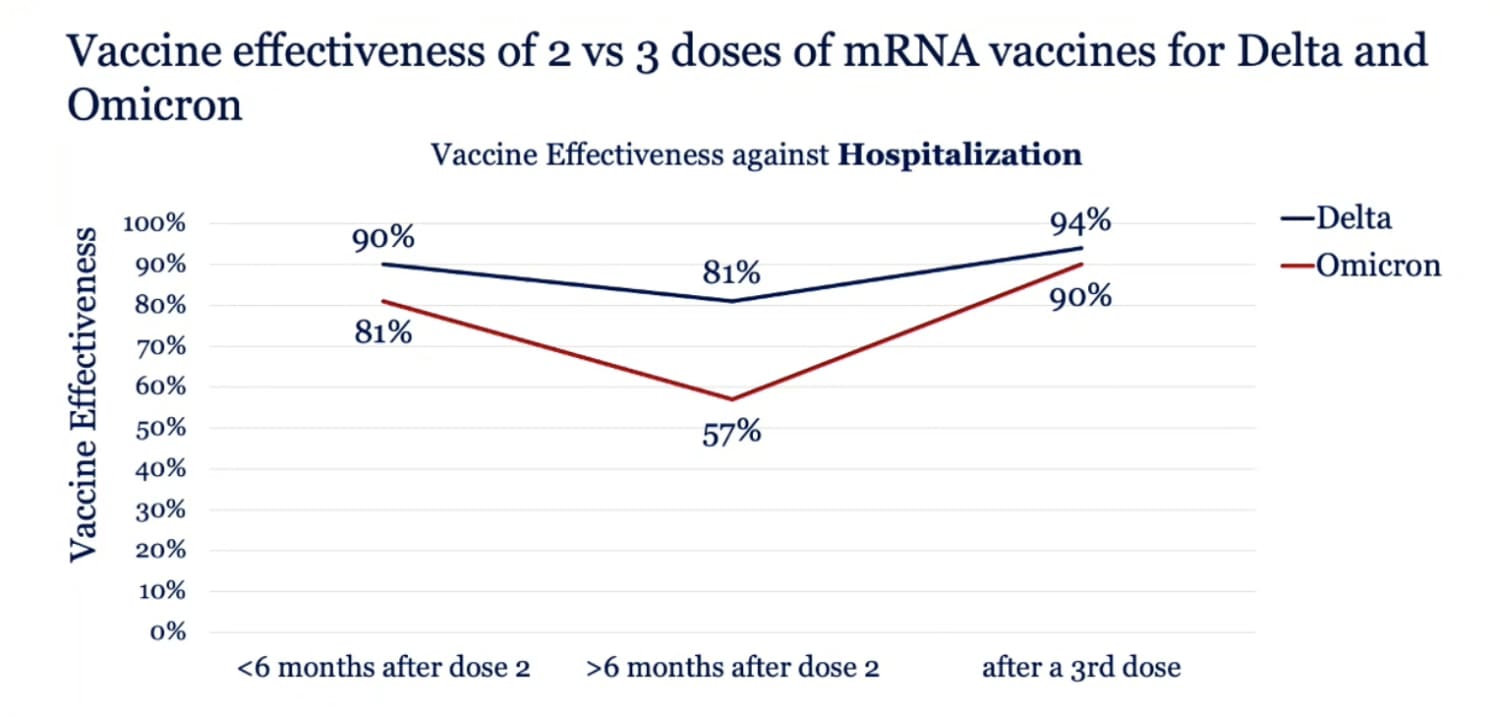
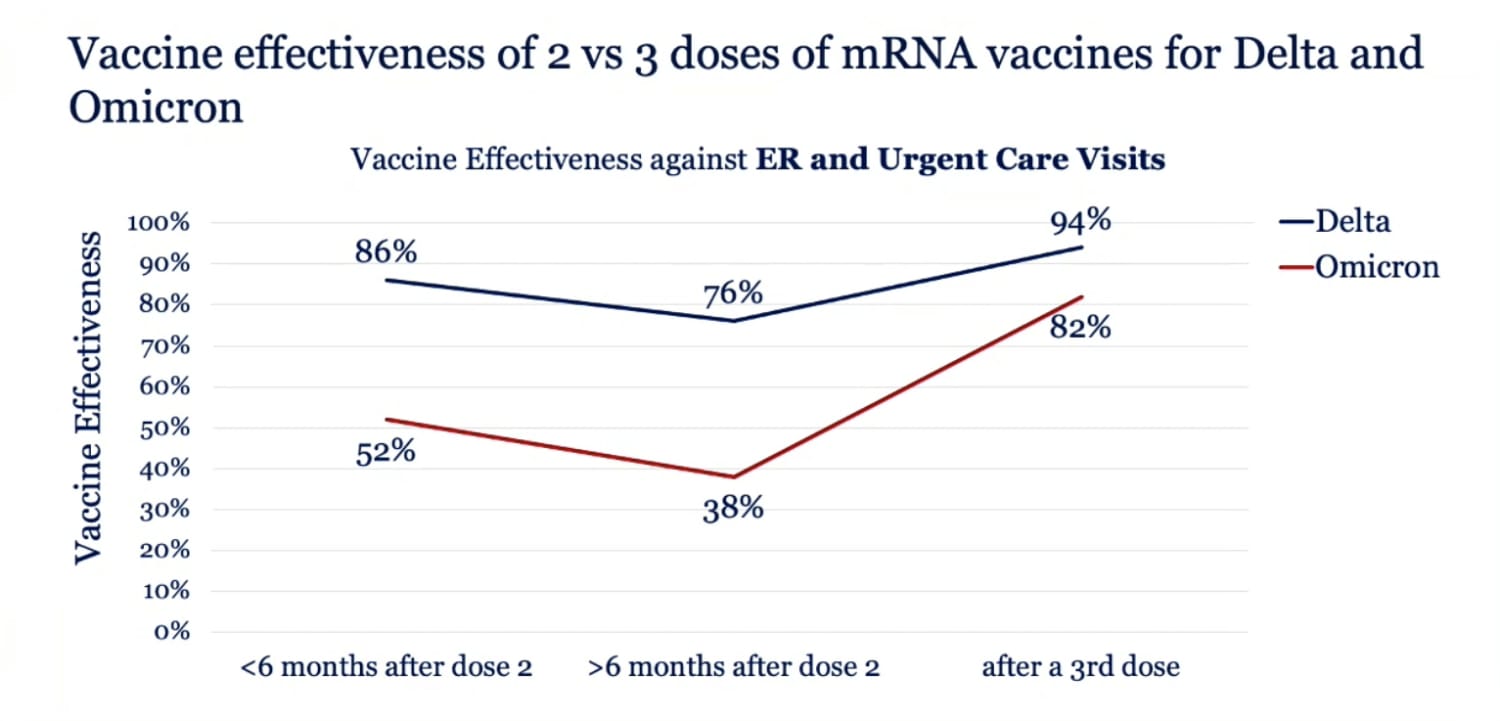
Effectiveness of a third dose of the BNT162b2 mRNA COVID-19 vaccine for preventing severe outcomes in Israel: an observational study - The Lancet

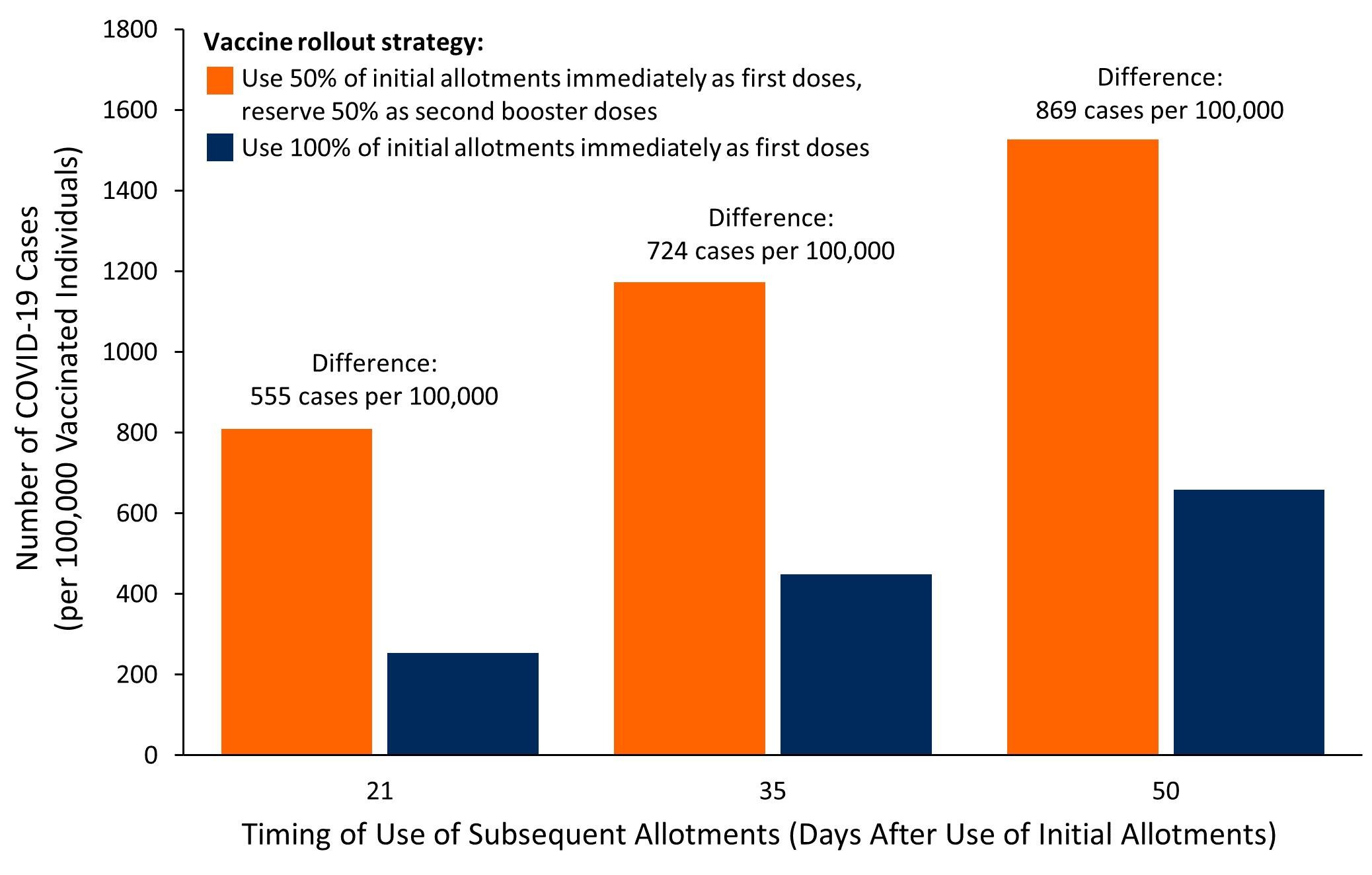
Coronavirus (COVID-19) Update: FDA Shortens Interval for Booster Dose of Moderna COVID-19 Vaccine to Five Months | FDA
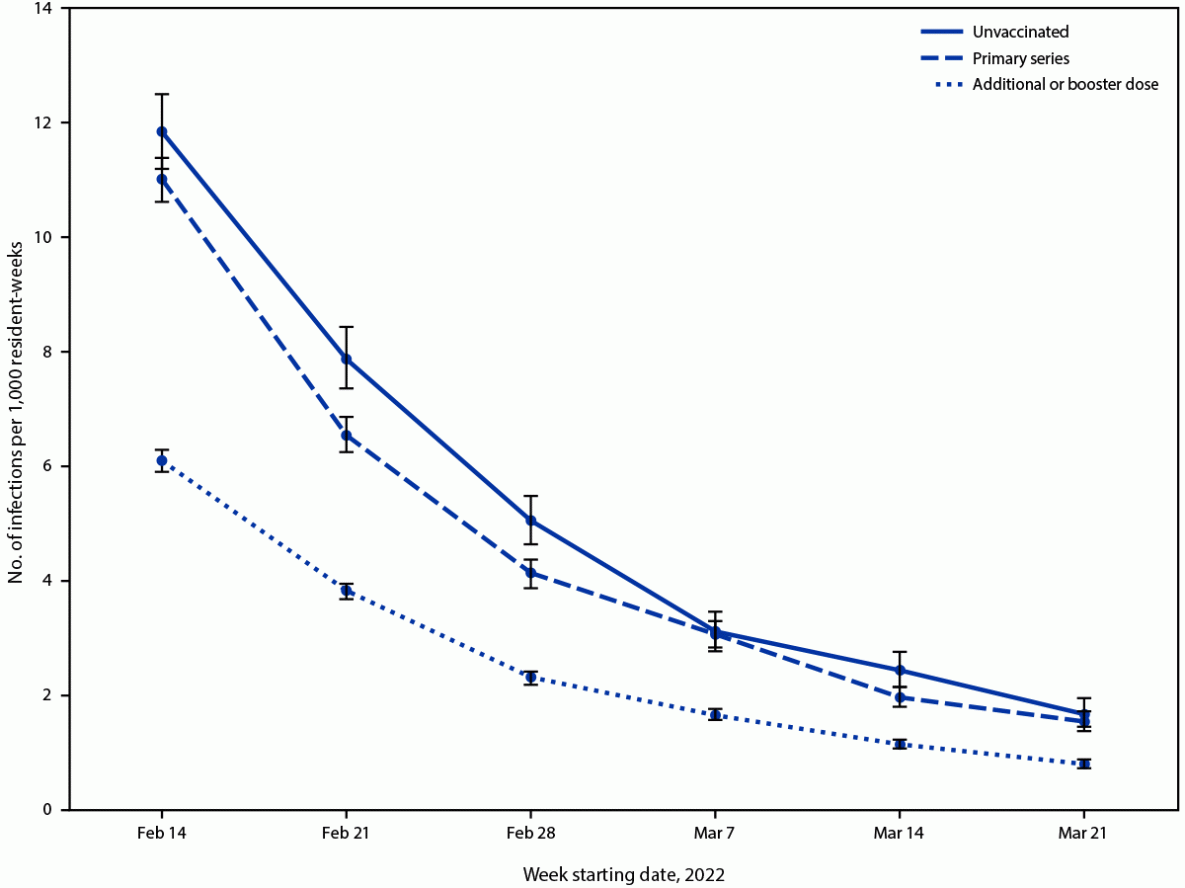
Effectiveness of a COVID-19 Additional Primary or Booster Vaccine Dose in Preventing SARS-CoV-2 Infection Among Nursing Home Residents During Widespread Circulation of the Omicron Variant — United States, February 14–March 27, 2022 | MMWR

Comparative Effectiveness and Antibody Responses to Moderna and Pfizer-BioNTech COVID-19 Vaccines among Hospitalized Veterans — Five Veterans Affairs Medical Centers, United States, February 1–September 30, 2021 | MMWR

Coronavirus (COVID-19) Update: FDA Takes Multiple Actions to Expand Use of Pfizer-BioNTech COVID-19 Vaccine | FDA

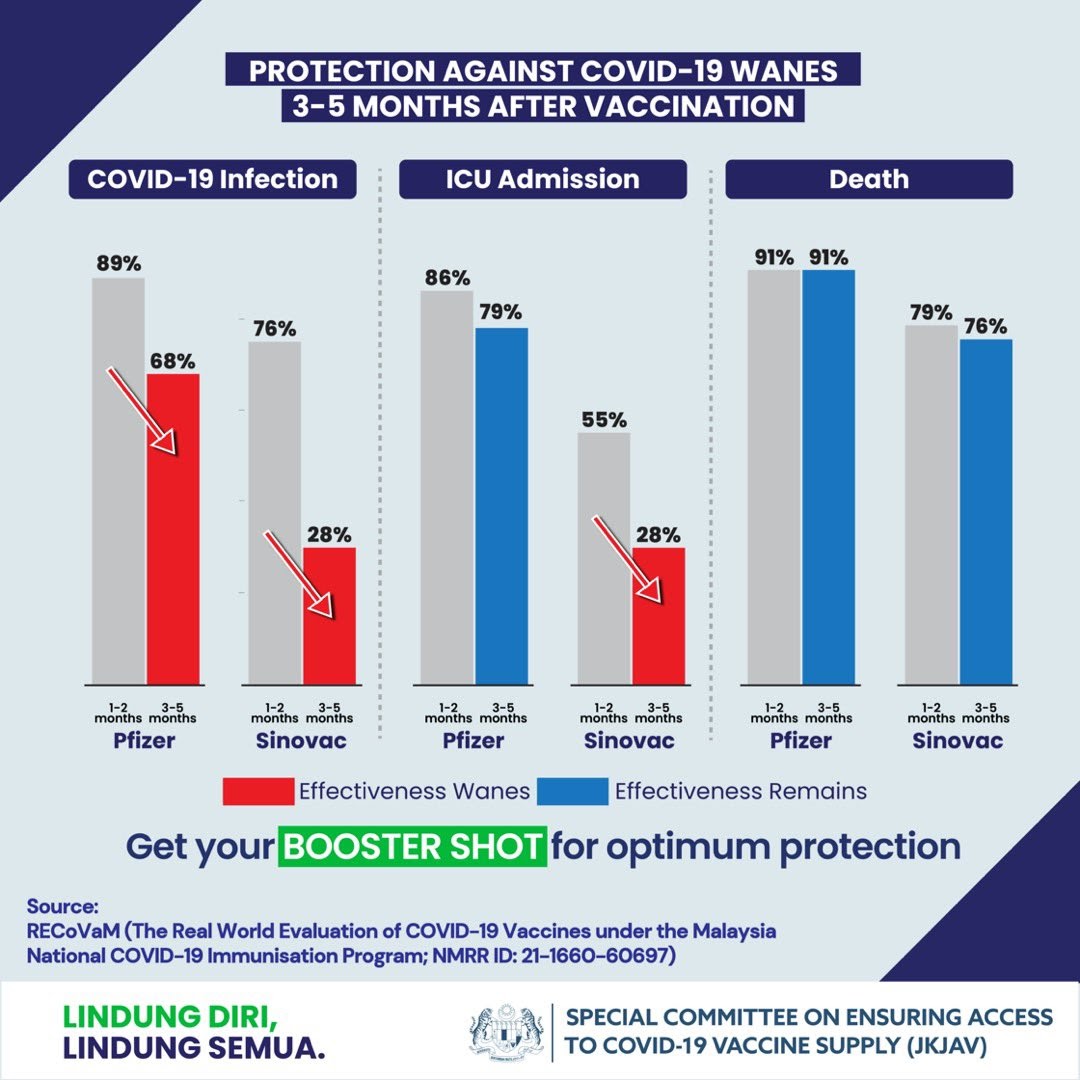
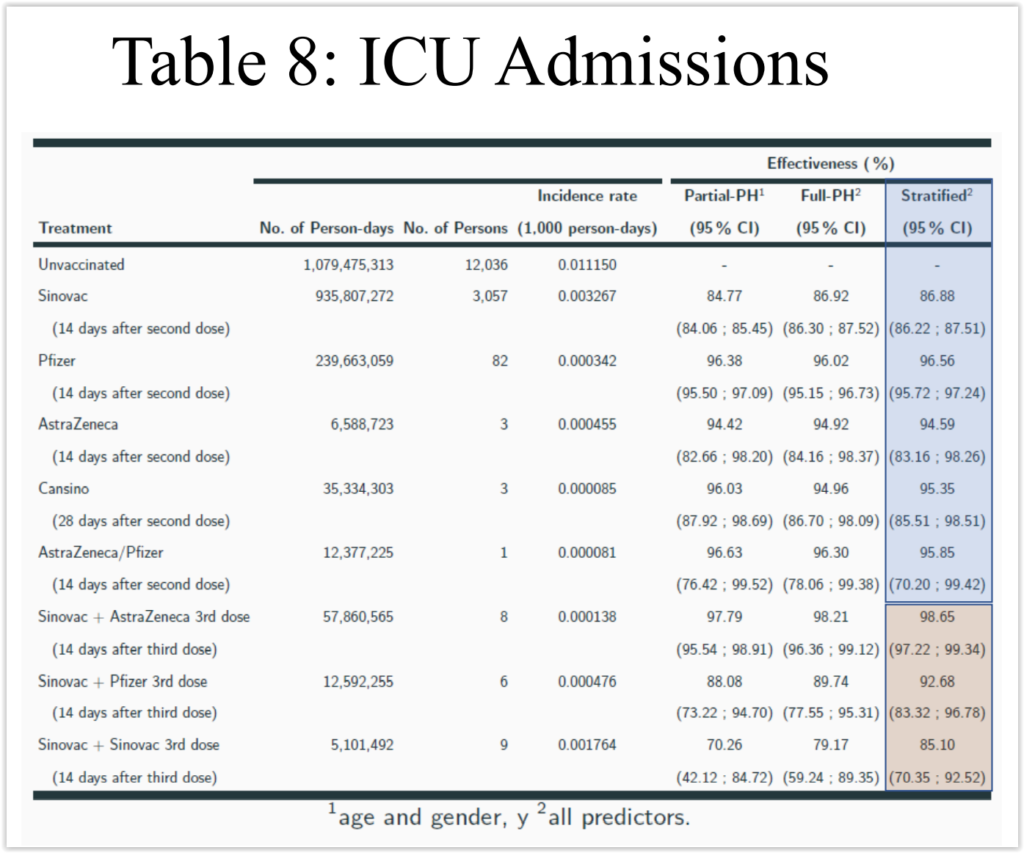
Effectiveness of an inactivated Covid-19 vaccine with homologous and heterologous boosters against Omicron in Brazil | Nature Communications

Khairy Jamaluddin 🇲🇾🌺 on X: "Real world data from Chile has shown significant increase in vaccine effectiveness after a booster for people who took Sinovac for their primary series. Biggest boost came

Effectiveness of mRNA BNT162b2 COVID-19 vaccine up to 6 months in a large integrated health system in the USA: a retrospective cohort study - The Lancet