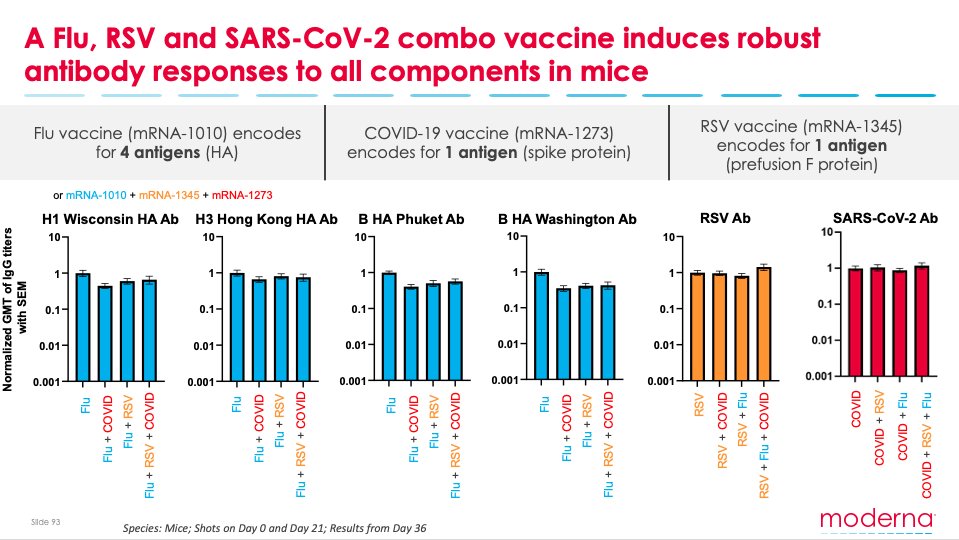
Moderna on X: "Today, we shared positive pre-clinical data demonstrating our ability to combine 6 mRNAs against 3 different respiratory viruses in 1 vaccine: COVID-19 booster + Flu booster + RSV booster. #

Variant SARS-CoV-2 mRNA vaccines confer broad neutralization as primary or booster series in mice - ScienceDirect
Study highlights the importance of bivalent mRNA booster vaccination in populations at high risk of severe COVID-19

Effectiveness of Bivalent mRNA Vaccines in Preventing Symptomatic SARS-CoV-2 Infection — Increasing Community Access to Testing Program, United States, September–November 2022 | MMWR

Early Estimates of Bivalent mRNA Booster Dose Vaccine Effectiveness in Preventing Symptomatic SARS-CoV-2 Infection Attributable to Omicron BA.5– and XBB/XBB.1.5–Related Sublineages Among Immunocompetent Adults — Increasing Community Access to Testing ...
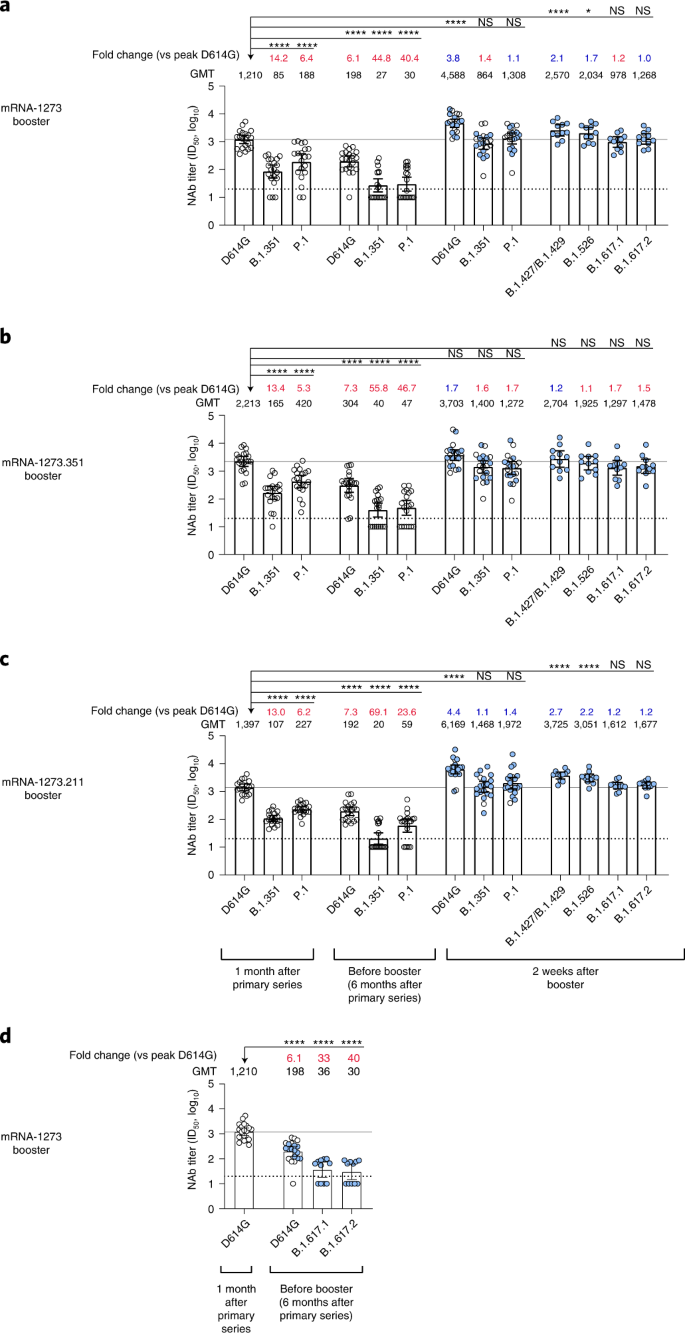
Safety and immunogenicity of SARS-CoV-2 variant mRNA vaccine boosters in healthy adults: an interim analysis | Nature Medicine

Moderna on X: "A 50 µg #booster dose of our first bivalent booster #vaccine candidate, mRNA-1273.211, demonstrated superiority against Beta, Delta and #Omicron variants of concern one month after administration, and superiority

Study: Booster dose of mRNA COVID vaccine prompts strong T cell response in immunocompromised patients

Safety of the fourth COVID-19 BNT162b2 mRNA (second booster) vaccine: a prospective and retrospective cohort study - The Lancet Respiratory Medicine

CDC - COVID-19 BOOSTER UPDATE: Everyone ages 18+ is eligible for a COVID-19 booster. If you got an mRNA vaccine, either Pfizer-BioNTech or Moderna, you may get a booster dose 6 months
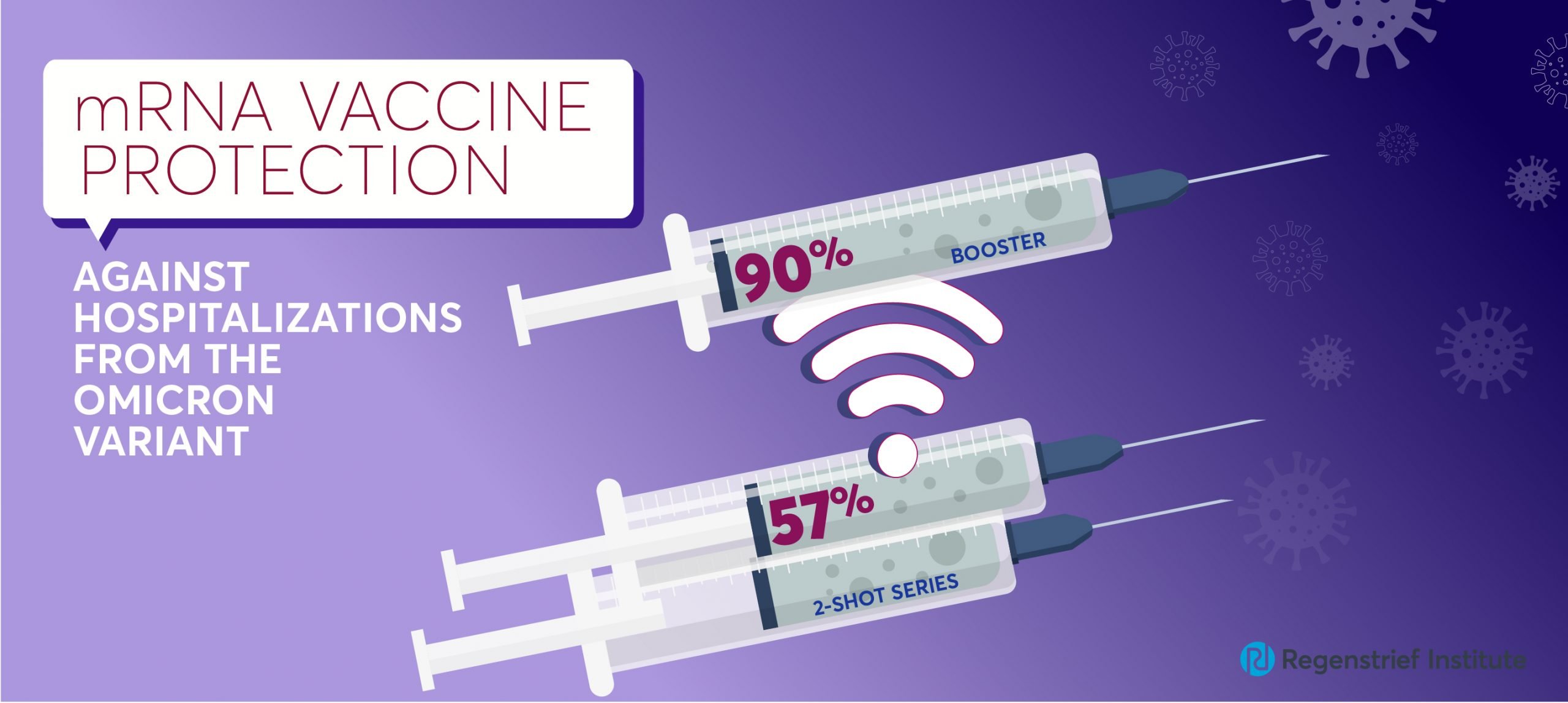
Three mRNA COVID-19 vaccine doses very effective in protecting against emergency department visits and hospitalizations associated with Omicron and Delta variants

Safety and immunogenicity of SARS-CoV-2 variant mRNA vaccine boosters in healthy adults: an interim analysis | Nature Medicine

Centers for Disease Control and Prevention on LinkedIn: CDC recommends Novavax's non-mRNA booster for people ages 18 and older…

Effectiveness of mRNA vaccine boosters against infection with the SARS-CoV-2 omicron (B.1.1.529) variant in Spain: a nationwide cohort study - The Lancet Infectious Diseases